# Numeric
x <- 1
y <- 3.14
z <- 1e4 # scientific notation
# Math operations
x + y[1] 4.14(x + 1) * y # follows PEMDAS[1] 6.28sqrt(y)[1] 1.772005log(y)[1] 1.144223y^2[1] 9.8596PLS 206 — Applied Multivariate Modeling
Grey Monroe
January 1, 2026
Everything in this course fits into one repeating cycle:
Collect data → Explore → Test / Model → CommunicateThis module covers the first two steps — loading data and exploring it — plus the classical hypothesis tests you’ll need when relationships are simple. Modules 2–10 build the modeling toolkit for when they’re not.
The single most important habit in data analysis: look at your data before you model it. Summary statistics can be identical across wildly different datasets (see Activity 3). Plot first, model second.
R is free, open-source, and used by ecologists, geneticists, agronomists, and data scientists worldwide. With 10,000+ packages on CRAN, it covers nearly every statistical method you’ll need. More importantly, your entire analysis lives in a script — reproducible, shareable, and tweakable.
This quarter we’ll use R for linear and generalized linear models, GAMs, regularization, PCA, clustering, random forests, neural nets, and SEM.
[1] 4.14[1] 6.28[1] 1.772005[1] 1.144223[1] 9.8596Use <- for assignment. Reserve = for function arguments only.
[1] "numeric"[1] "character"[1] "logical"[1] TRUE[1] TRUE[1] FALSER will silently coerce types when you combine mismatched data — a common source of bugs when reading CSVs. Always check with class() or str() after import.
[1] "character"[1] 3.14[1] 1 2 3[1] 10 2 30Never convert a factor directly to numeric. Always go as.numeric(as.character(x)).
Everything in R is a vector. A single number is a vector of length 1.
[1] 2 4 6 8 10 20[1] 4 5 10[1] "banana"[1] 4 5 10[1] 4 5 10[1] TRUE FALSE FALSE TRUE[1] "apple" "grape"[1] FALSE FALSE TRUE FALSE[1] NA[1] 2.333333[1] "apple" "banana" "cherry"fruits
apple banana cherry
2 1 1 [1] 42[1] "hello"'data.frame': 4 obs. of 3 variables:
$ rep : num 1 2 3 4
$ treat: chr "A" "B" "A" "B"
$ value: num 10.2 8.5 11.1 9.3[1] 10.2 8.5 11.1 9.3 rep treat value
1 1 A 10.2 rep treat value
1 1 A 10.2
3 3 A 11.1 rep treat value
1 1 A 10.2
2 2 B 8.5
3 3 A 11.1
4 4 B 9.3 rep treat value
1 1 A 10.2
2 2 B 8.5
3 3 A 11.1
4 4 B 9.3 rep treat value
1 1 A 10.2
2 2 B 8.5
3 3 A 11.1
4 4 B 9.3 rep treat value log_value
Min. :1.00 Length:4 Min. : 8.500 Min. :2.140
1st Qu.:1.75 Class :character 1st Qu.: 9.100 1st Qu.:2.208
Median :2.50 Mode :character Median : 9.750 Median :2.276
Mean :2.50 Mean : 9.775 Mean :2.275
3rd Qu.:3.25 3rd Qu.:10.425 3rd Qu.:2.344
Max. :4.00 Max. :11.100 Max. :2.407 'data.frame': 178 obs. of 14 variables:
$ Cultivar : chr "barolo" "barolo" "barolo" "barolo" ...
$ Alcohol : num 14.2 13.2 13.2 14.4 13.2 ...
$ AlcAsh : num 15.6 11.2 18.6 16.8 21 15.2 14.6 17.6 14 16 ...
$ Ash : num 2.43 2.14 2.67 2.5 2.87 2.45 2.45 2.61 2.17 2.27 ...
$ Color : num 5.64 4.38 5.68 7.8 4.32 6.75 5.25 5.05 5.2 7.22 ...
$ Flav : num 3.06 2.76 3.24 3.49 2.69 3.39 2.52 2.51 2.98 3.15 ...
$ Hue : num 1.04 1.05 1.03 0.86 1.04 1.05 1.02 1.06 1.08 1.01 ...
$ MalicAcid : num 1.71 1.78 2.36 1.95 2.59 1.76 1.87 2.15 1.64 1.35 ...
$ Mg : int 127 100 101 113 118 112 96 121 97 98 ...
$ NonFlavPhenols: num 0.28 0.26 0.3 0.24 0.39 0.34 0.3 0.31 0.29 0.22 ...
$ OD : num 3.92 3.4 3.17 3.45 2.93 2.85 3.58 3.58 2.85 3.55 ...
$ Phenols : num 2.8 2.65 2.8 3.85 2.8 3.27 2.5 2.6 2.8 2.98 ...
$ Proa : num 2.29 1.28 2.81 2.18 1.82 1.97 1.98 1.25 1.98 1.85 ...
$ Proline : int 1065 1050 1185 1480 735 1450 1290 1295 1045 1045 ... Cultivar Alcohol AlcAsh Ash Color Flav Hue MalicAcid Mg NonFlavPhenols
1 barolo 14.23 15.6 2.43 5.64 3.06 1.04 1.71 127 0.28
2 barolo 13.20 11.2 2.14 4.38 2.76 1.05 1.78 100 0.26
3 barolo 13.16 18.6 2.67 5.68 3.24 1.03 2.36 101 0.30
4 barolo 14.37 16.8 2.50 7.80 3.49 0.86 1.95 113 0.24
5 barolo 13.24 21.0 2.87 4.32 2.69 1.04 2.59 118 0.39
6 barolo 14.20 15.2 2.45 6.75 3.39 1.05 1.76 112 0.34
OD Phenols Proa Proline
1 3.92 2.80 2.29 1065
2 3.40 2.65 1.28 1050
3 3.17 2.80 2.81 1185
4 3.45 3.85 2.18 1480
5 2.93 2.80 1.82 735
6 2.85 3.27 1.97 1450Always run str() after reading data — verify that column types match what you expect (e.g., numeric not character).
You will see |> (base R pipe, R ≥ 4.1) and %>% (magrittr, from the tidyverse) constantly in R code online. They both mean the same thing: pass the left-hand side as the first argument to the right-hand side.
[1] 2[1] 2Pipes shine when you chain multiple operations:
[1] 4[1] 4|> is built into R ≥ 4.1 — no packages needed. %>% requires library(magrittr) or library(dplyr). They behave identically in most cases. Prefer |> in new code.
When you need to do the same operation across many columns or groups, reach for apply() or sapply() instead of copy-pasting code. This will come up immediately in the problem set.
Sepal.Length Sepal.Width Petal.Length Petal.Width
5.843333 3.057333 3.758000 1.199333 Sepal.Length Sepal.Width Petal.Length Petal.Width
0.8280661 0.4358663 1.7652982 0.7622377 Sepal.Length mean = 5.843333
Sepal.Width mean = 3.057333
Petal.Length mean = 3.758
Petal.Width mean = 1.199333 More on apply(), lapply(), and writing your own functions comes in Module 2. For now, the pattern is: apply(data_frame, 2, some_function).
ggplot2 builds plots layer by layer:
ggplot(data, aes(x = ..., y = ...)) +
geom_*() + # geometry: points, lines, bars, histograms ...
labs() + # labels
theme_*() # stylingEvery plot starts with ggplot(). Add layers with +.
The first thing to do with any new variable — look at its distribution.
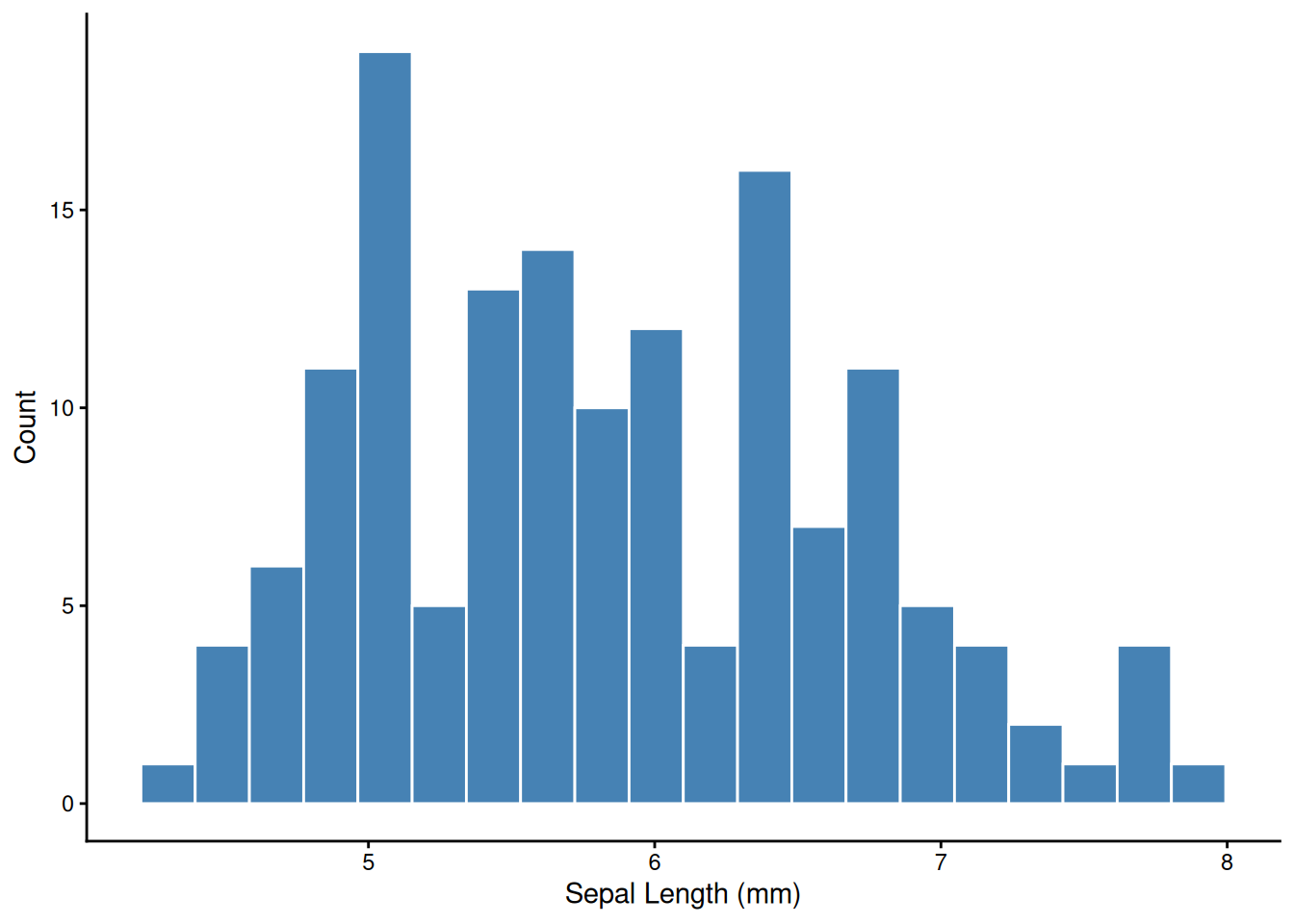
facet_wrap() splits one plot into panels by a grouping variable — one of ggplot’s most powerful tools for exploratory analysis.
ggplot(iris,
aes(x = Sepal.Length, y = Sepal.Width,
col = Species, fill = Species)) +
geom_point(alpha = 0.7) +
geom_smooth(method = "lm", alpha = 0.1) +
labs(title = "Iris Dataset",
x = "Sepal Length (mm)",
y = "Sepal Width (mm)") +
scale_color_manual(values = c("gray30", "dodgerblue", "orange")) +
scale_fill_manual(values = c("gray30", "dodgerblue", "orange")) +
theme_classic(base_size = 14) +
theme(plot.title = element_text(face = "bold"),
legend.position = "top")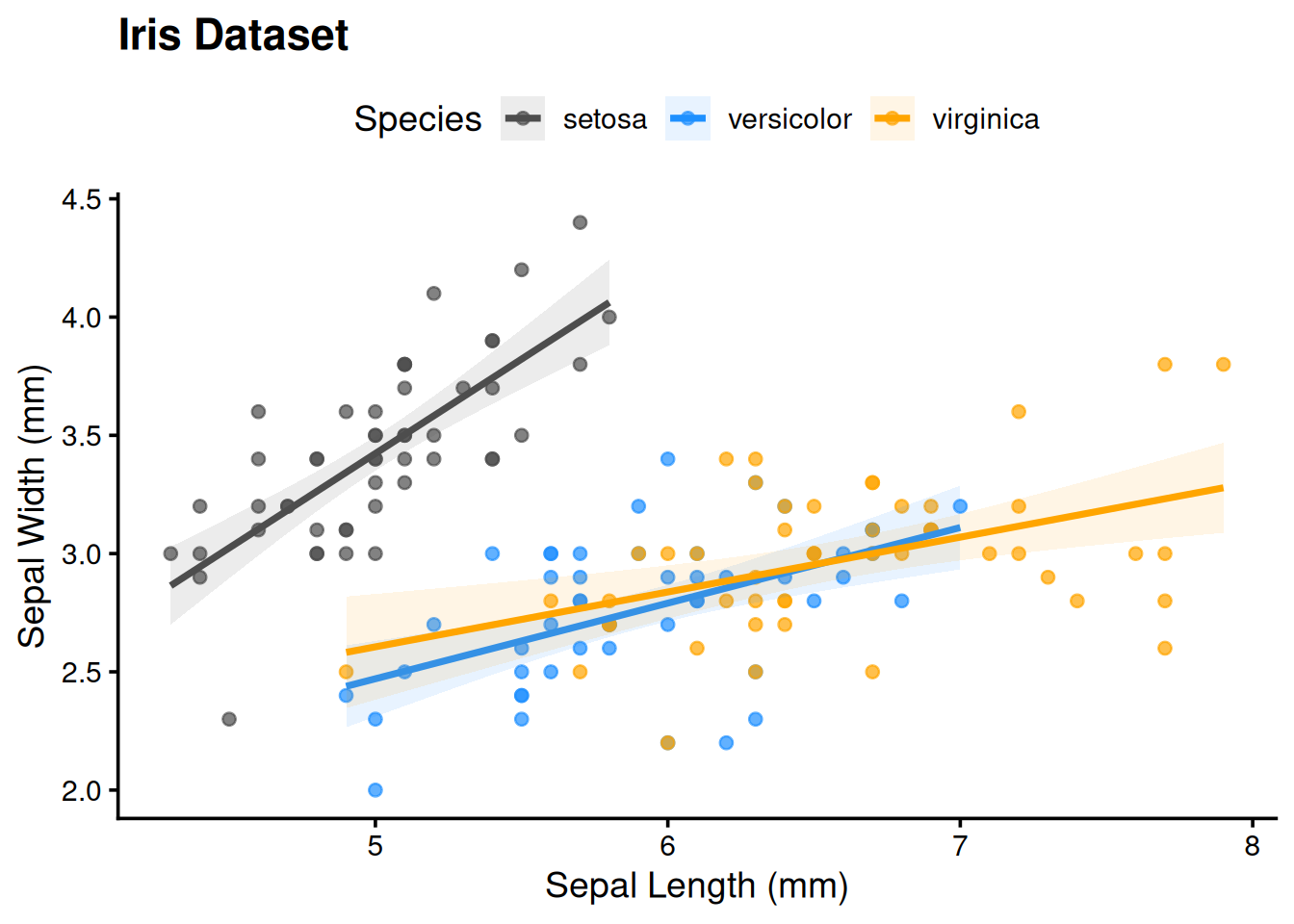
Use ggsave() rather than screenshot. It gives you a reproducible, high-resolution file at any dimension you need.
The GGally package extends ggplot2 with ggpairs() — a scatter plot matrix showing pairwise relationships, distributions, and correlations in one figure. You’ll use it in the problem set:
Before running any tests, anchor the key concepts.
A p-value is the probability of observing a result at least this extreme if the null hypothesis were true. It is not:
p < 0.05 does not mean the effect is real or important. With n = 10,000, a trivially small effect will be significant. Always report both a p-value and an effect size.
| H₀ is actually true | H₀ is actually false | |
|---|---|---|
| Reject H₀ (p < α) | Type I error (false positive) — rate = α | Correct (true positive) — power = 1 − β |
| Fail to reject H₀ | Correct (true negative) | Type II error (false negative) — rate = β |
Setting α = 0.05 means you accept a 5% chance of a false positive per test — which becomes a serious problem when running many tests (see Multiple Comparisons below).
Statistical power (1 − β) is the probability of detecting a real effect. Power increases with larger sample size, larger effect size, and lower measurement noise. See the t-test Explorer to watch power change in real time.
| Outcome | Groups / structure | Normally distributed? | Test |
|---|---|---|---|
| Continuous | 1 group vs. fixed value | Yes | One-sample t-test |
| Continuous | 1 group vs. fixed value | No | Wilcoxon signed-rank |
| Continuous | 2 independent groups | Yes | Welch’s two-sample t-test |
| Continuous | 2 independent groups | No | Mann-Whitney U |
| Continuous | 2 paired / matched groups | Yes | Paired t-test |
| Continuous | 2 paired / matched groups | No | Wilcoxon signed-rank (paired) |
| Continuous | ≥ 3 independent groups | Yes | One-way ANOVA + TukeyHSD |
| Continuous | ≥ 3 independent groups | No | Kruskal-Wallis + pairwise Wilcoxon |
| Continuous | 2 continuous variables | Linear | Pearson correlation |
| Continuous | 2 continuous variables | Monotone / outliers | Spearman correlation |
| Categorical | 2 × 2 table, small n | — | Fisher’s exact |
| Categorical | 2 × 2 or larger, large n | — | Chi-squared |
Normality refers to your residuals, not your raw data. For small samples (n < 30), check with shapiro.test() and a QQ plot. For large samples, the central limit theorem means most tests are robust even with non-normal data.
$ctrl
Min. 1st Qu. Median Mean 3rd Qu. Max.
4.170 4.550 5.155 5.032 5.293 6.110
$trt1
Min. 1st Qu. Median Mean 3rd Qu. Max.
3.590 4.207 4.550 4.661 4.870 6.030
$trt2
Min. 1st Qu. Median Mean 3rd Qu. Max.
4.920 5.268 5.435 5.526 5.735 6.310 Does the control group mean differ from 5.0 g?
One Sample t-test
data: ctrl
t = 0.17355, df = 9, p-value = 0.8661
alternative hypothesis: true mean is not equal to 5
95 percent confidence interval:
4.614882 5.449118
sample estimates:
mean of x
5.032 Non-parametric alternative:
Welch Two Sample t-test
data: x1 and x2
t = 1.1913, df = 16.524, p-value = 0.2504
alternative hypothesis: true difference in means is not equal to 0
95 percent confidence interval:
-0.2875162 1.0295162
sample estimates:
mean of x mean of y
5.032 4.661
Wilcoxon rank sum test with continuity correction
data: x1 and x2
W = 67.5, p-value = 0.1986
alternative hypothesis: true location shift is not equal to 0
95 percent confidence interval:
-0.2899731 1.0100554
sample estimates:
difference in location
0.4213948 Welch’s t-test is the safe default — it doesn’t assume equal variances between groups.
Use a paired t-test when each observation in group A has a natural match in group B — before/after measurements on the same subject, plots receiving both treatments, twin studies. Pairing removes between-subject variation and increases power.
Paired t-test
data: post and pre
t = 3.0348, df = 11, p-value = 0.01135
alternative hypothesis: true mean difference is not equal to 0
95 percent confidence interval:
0.4714789 2.9607031
sample estimates:
mean difference
1.716091
One Sample t-test
data: post - pre
t = 3.0348, df = 11, p-value = 0.01135
alternative hypothesis: true mean is not equal to 0
95 percent confidence interval:
0.4714789 2.9607031
sample estimates:
mean of x
1.716091 # Visualize paired structure
plot_df <- data.frame(
plot = rep(1:12, 2),
time = rep(c("Pre", "Post"), each = 12),
nitrate = c(pre, post)
)
plot_df$time <- factor(plot_df$time, levels = c("Pre", "Post"))
ggplot(plot_df, aes(x = time, y = nitrate, group = plot)) +
geom_line(alpha = 0.5) +
geom_point(size = 2) +
labs(x = NULL, y = "Nitrate (mg/kg)",
title = "Paired before/after measurements") +
theme_classic()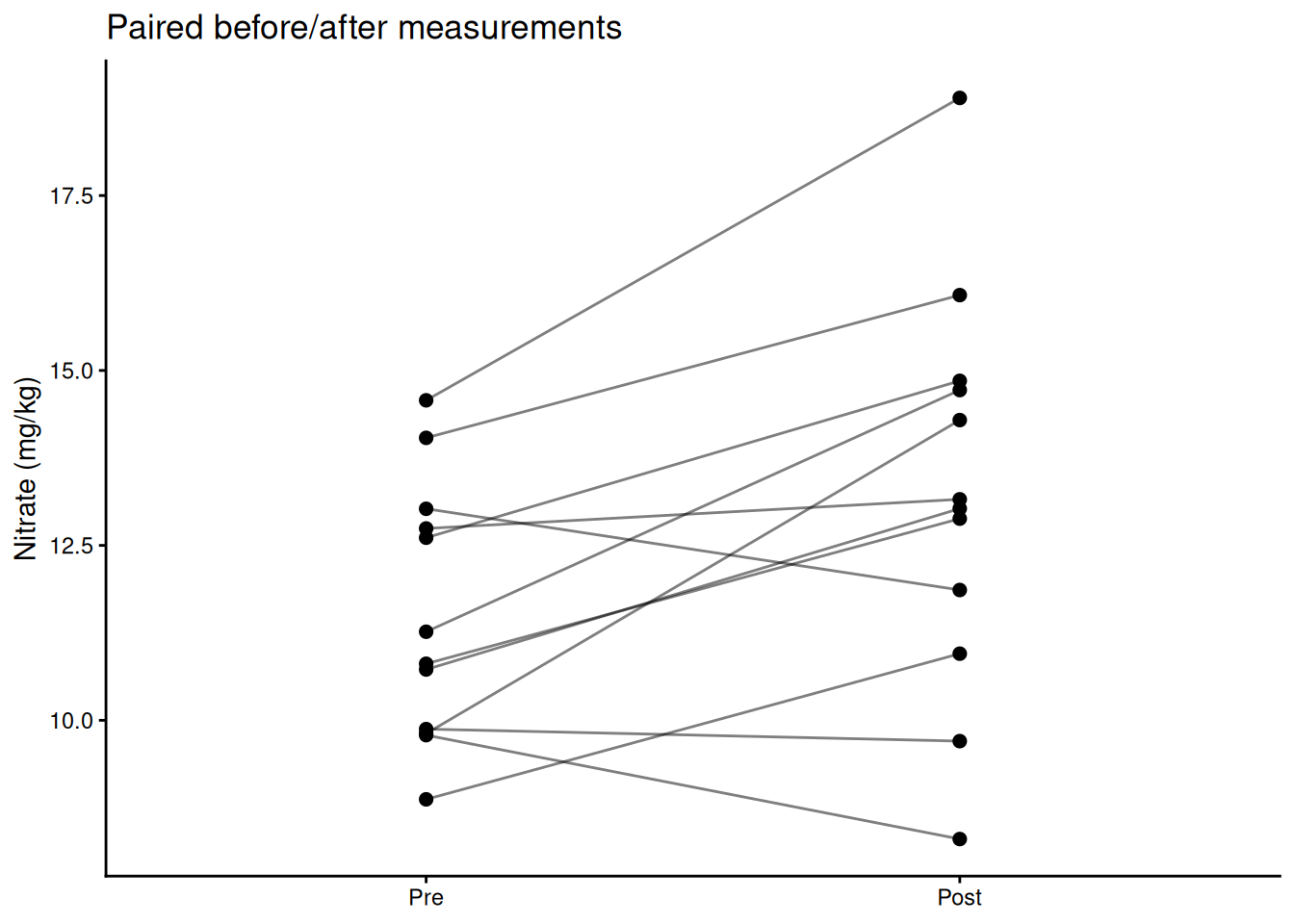
Non-parametric alternative:
Wilcoxon signed rank test with continuity correction
data: post and pre
V = 70, p-value = 0.01673
alternative hypothesis: true location shift is not equal to 0
95 percent confidence interval:
0.4077382 3.2019135
sample estimates:
(pseudo)median
2.045461 A significant p-value tells you the effect probably isn’t zero. Effect size tells you how big it is — which is what actually matters for biology.
\[d = \frac{|\bar{x}_1 - \bar{x}_2|}{s_\text{pooled}}\]
[1] 0.5327478| d | Interpretation |
|---|---|
| < 0.2 | Negligible |
| 0.2 – 0.5 | Small |
| 0.5 – 0.8 | Medium |
| > 0.8 | Large |
η² is the proportion of total variance explained by the group factor — analogous to R² in regression.
[1] 0.2641483Report effect sizes in your results. A biologically trivial effect can be highly significant with a large enough sample. A large effect can be non-significant with a small sample. Neither alone is the whole story.
Do all three treatment groups differ?
Df Sum Sq Mean Sq F value Pr(>F)
group 2 3.766 1.8832 4.846 0.0159 *
Residuals 27 10.492 0.3886
---
Signif. codes: 0 '***' 0.001 '**' 0.01 '*' 0.05 '.' 0.1 ' ' 1Post-hoc pairwise comparisons:
Tukey multiple comparisons of means
95% family-wise confidence level
Fit: aov(formula = weight ~ group, data = PlantGrowth)
$group
diff lwr upr p adj
trt1-ctrl -0.371 -1.0622161 0.3202161 0.3908711
trt2-ctrl 0.494 -0.1972161 1.1852161 0.1979960
trt2-trt1 0.865 0.1737839 1.5562161 0.0120064ggplot(PlantGrowth, aes(group, weight, fill = group)) +
geom_boxplot(alpha = 0.75, show.legend = FALSE) +
geom_jitter(width = 0.08, alpha = 0.6) +
labs(title = "Raw values (ANOVA)") +
theme_classic()
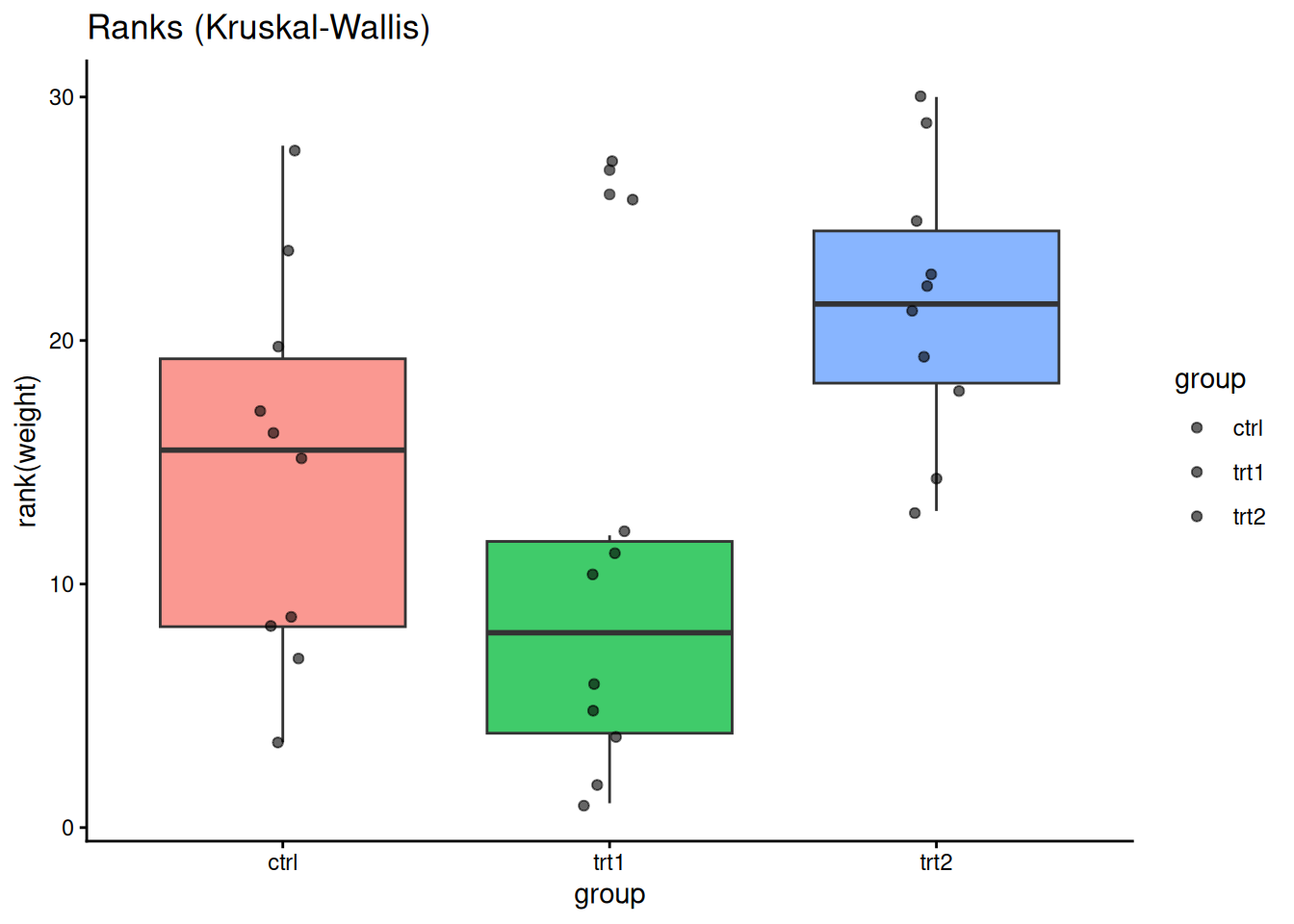
ggplot(PlantGrowth, aes(group, rank(weight), fill = group)) +
geom_boxplot(alpha = 0.75, show.legend = FALSE) +
geom_jitter(width = 0.08, alpha = 0.6) +
labs(title = "Ranks (Kruskal-Wallis)") +
theme_classic()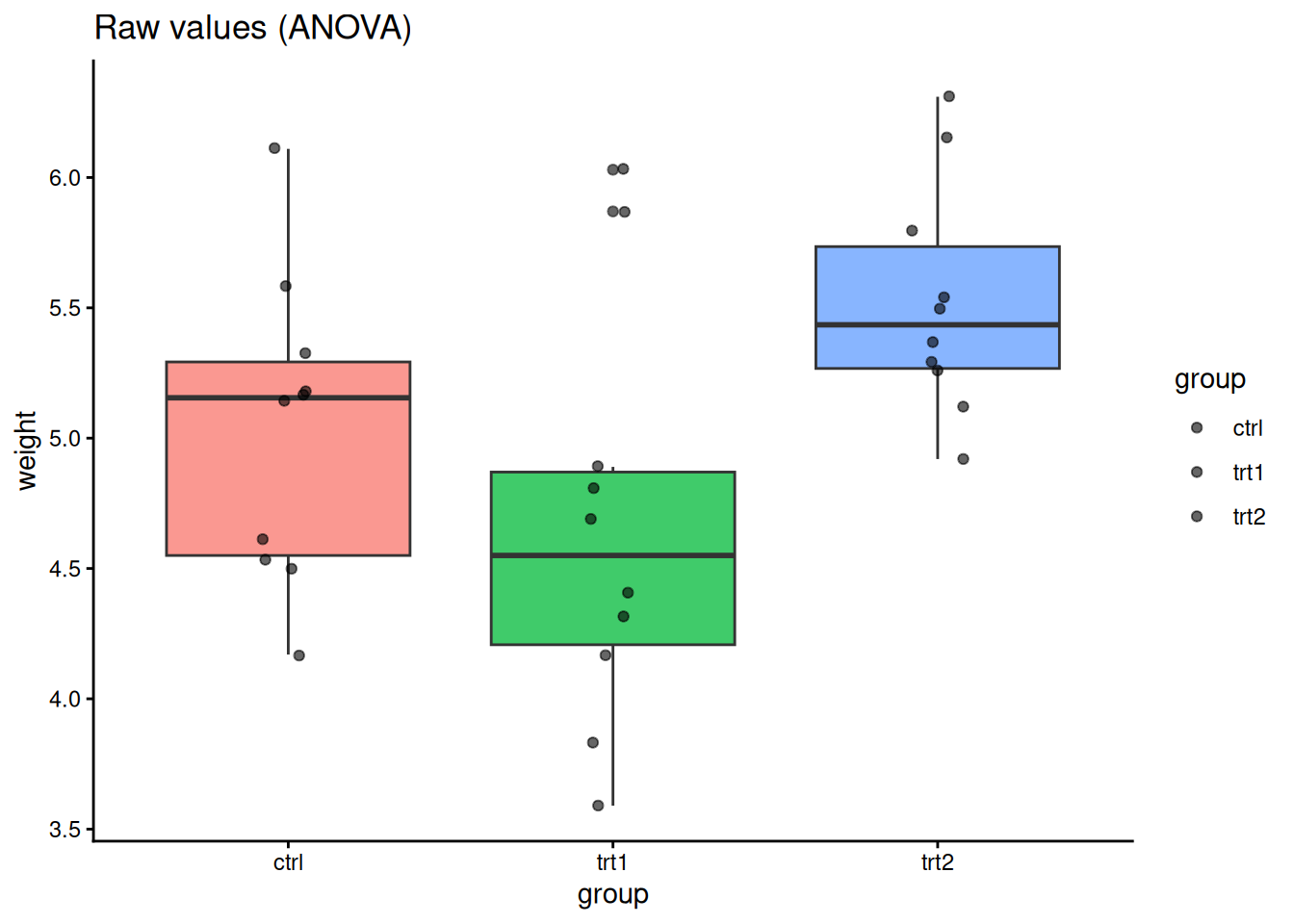
Every statistical test you run has a α = 0.05 chance of a false positive. If you run 20 independent tests, you expect one false positive on average — not because the biology changed, but because of random chance.
The problem: computing 10 pairwise correlations between wine variables gives you 10 chances for a spurious significant result. Running ANOVA post-hoc tests compounds the error further.
The solution: adjust p-values upward to account for the number of tests.
Common methods, roughly in order of conservatism:
pairwise.wilcox.test, p.adjust) — good general choice, less conservative than Bonferroni
Pairwise comparisons using Wilcoxon rank sum test with continuity correction
data: PlantGrowth$weight and PlantGrowth$group
ctrl trt1
trt1 0.199 -
trt2 0.126 0.027
P value adjustment method: holm [1] 0.040 0.090 0.090 0.200 0.025[1] 0.025 0.050 0.050 0.200 0.025
Pearson's product-moment correlation
data: sub$conc and sub$uptake
t = 4.3196, df = 19, p-value = 0.0003695
alternative hypothesis: true correlation is not equal to 0
95 percent confidence interval:
0.3910239 0.8709363
sample estimates:
cor
0.7038936
Spearman's rank correlation rho
data: sub$conc and sub$uptake
S = 256.28, p-value = 2.691e-06
alternative hypothesis: true rho is not equal to 0
sample estimates:
rho
0.8335869 ggplot(sub, aes(conc, uptake)) +
geom_point() +
geom_smooth(method = "lm", linewidth = 0.8) +
geom_smooth(method = "loess", linetype = 2, linewidth = 0.8) +
labs(title = "Raw", x = "CO2 concentration", y = "Uptake") +
theme_classic()
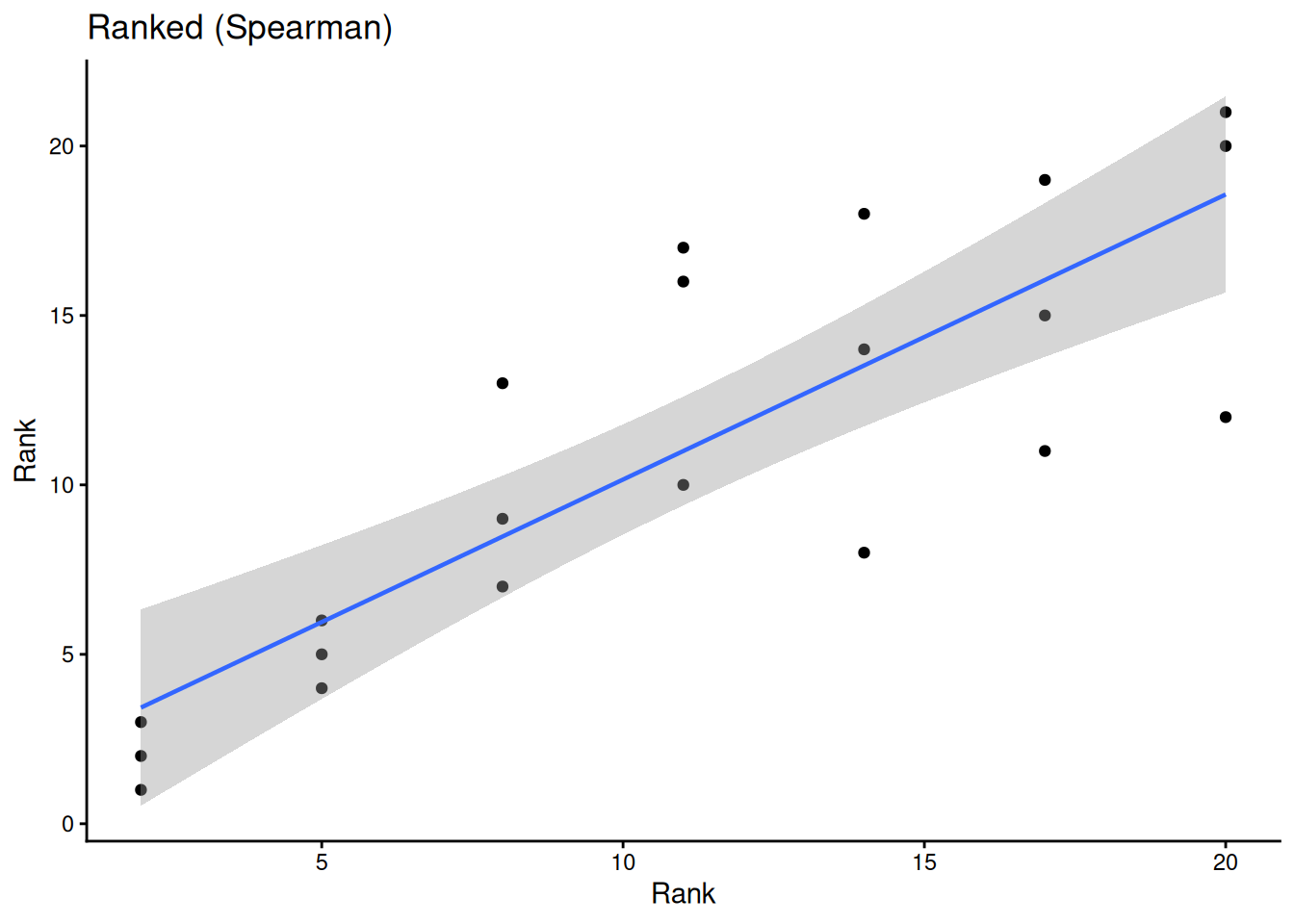
ggplot(sub, aes(rank(conc), rank(uptake))) +
geom_point() +
geom_smooth(method = "lm", linewidth = 0.8) +
labs(title = "Ranked (Spearman)", x = "Rank", y = "Rank") +
theme_classic()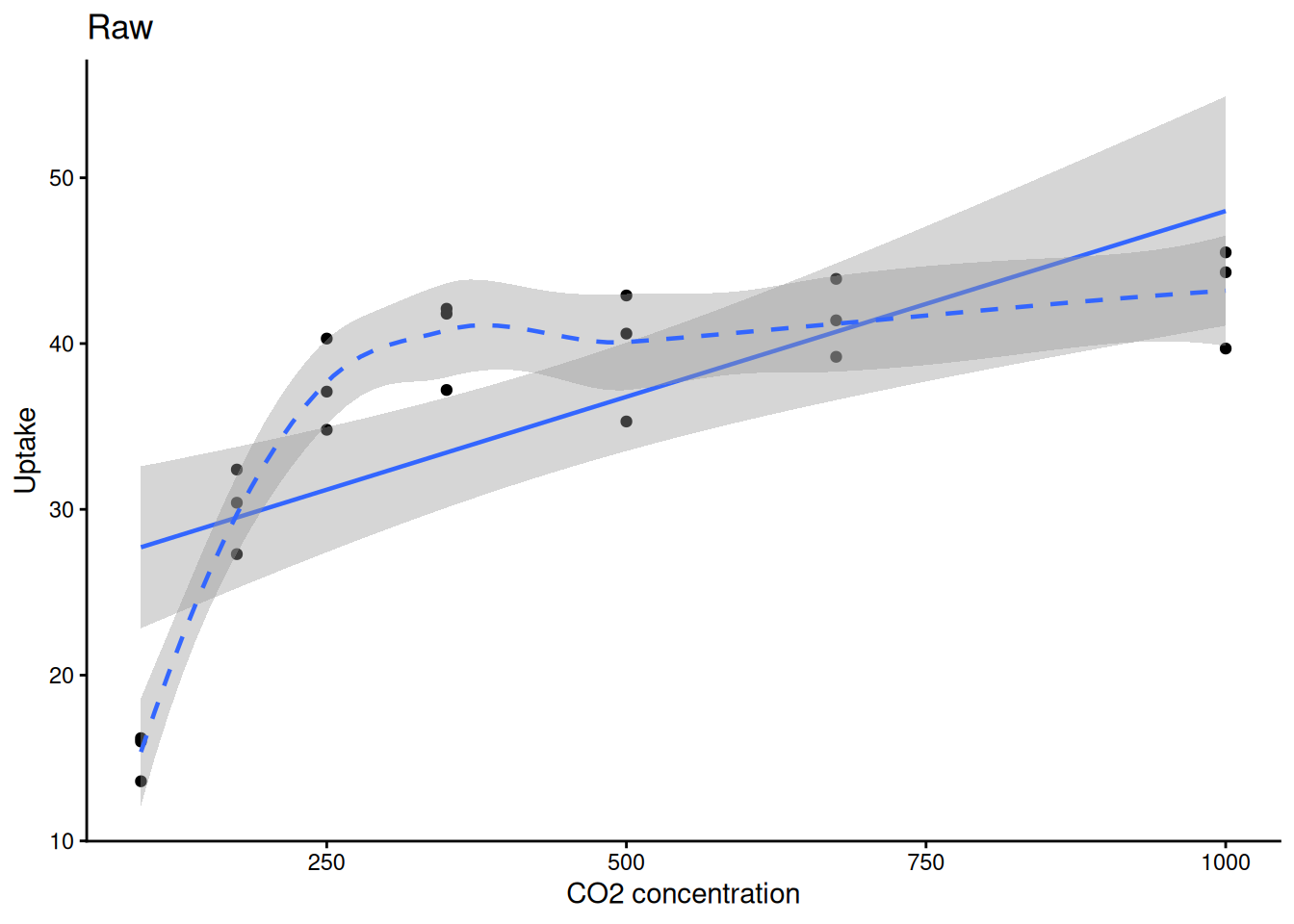
Is pest presence associated with treatment?
Trt
Pest A B
Present 3 17
Absent 10 10
Fisher's Exact Test for Count Data
data: tab
p-value = 0.04074
alternative hypothesis: true odds ratio is not equal to 1
95 percent confidence interval:
0.02632766 0.94457354
sample estimates:
odds ratio
0.184812
Pearson's Chi-squared test
data: tab
X-squared = 5.584, df = 1, p-value = 0.01812Use Fisher’s exact when any cell count is < 5. Use chi-squared for larger samples.
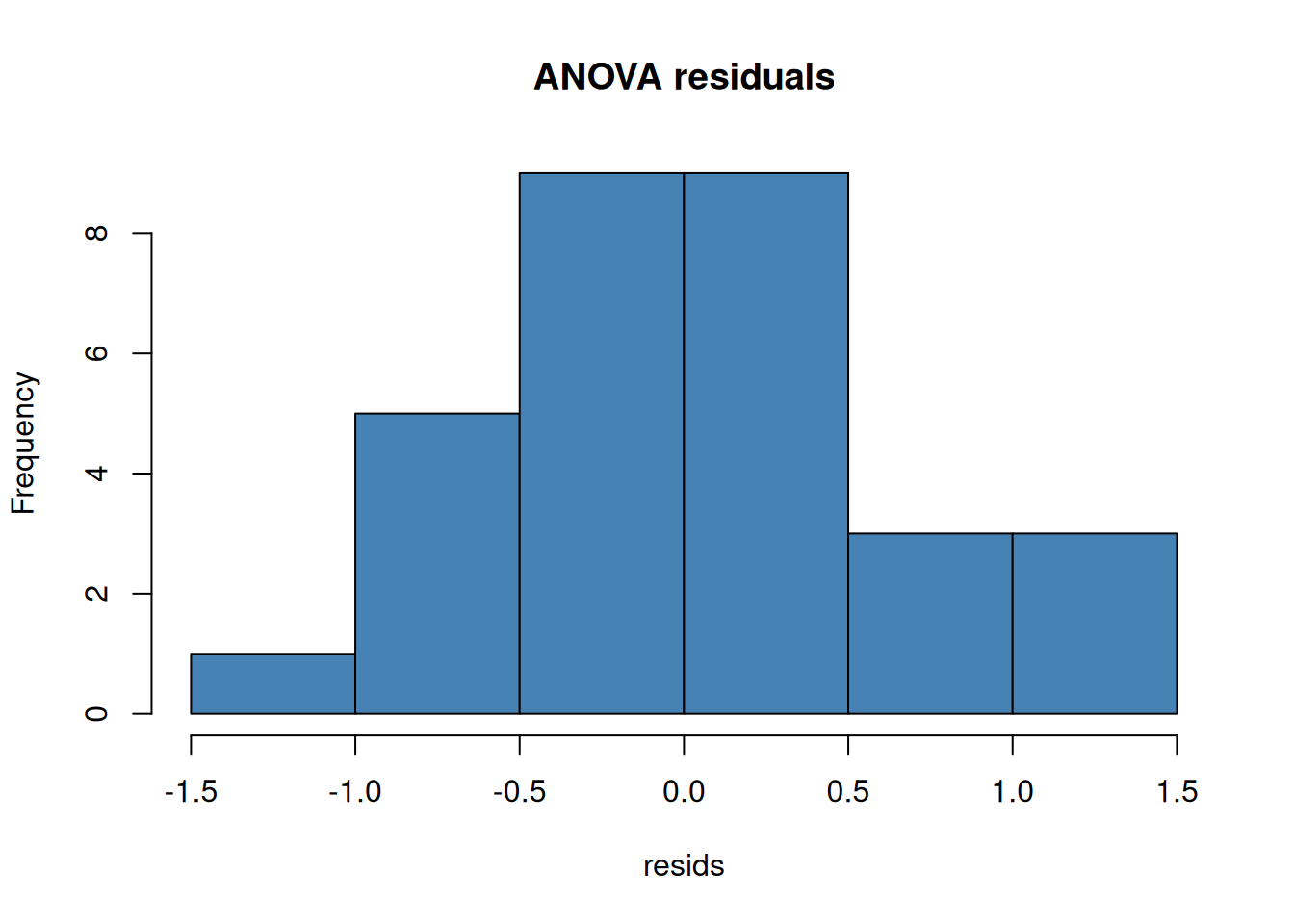
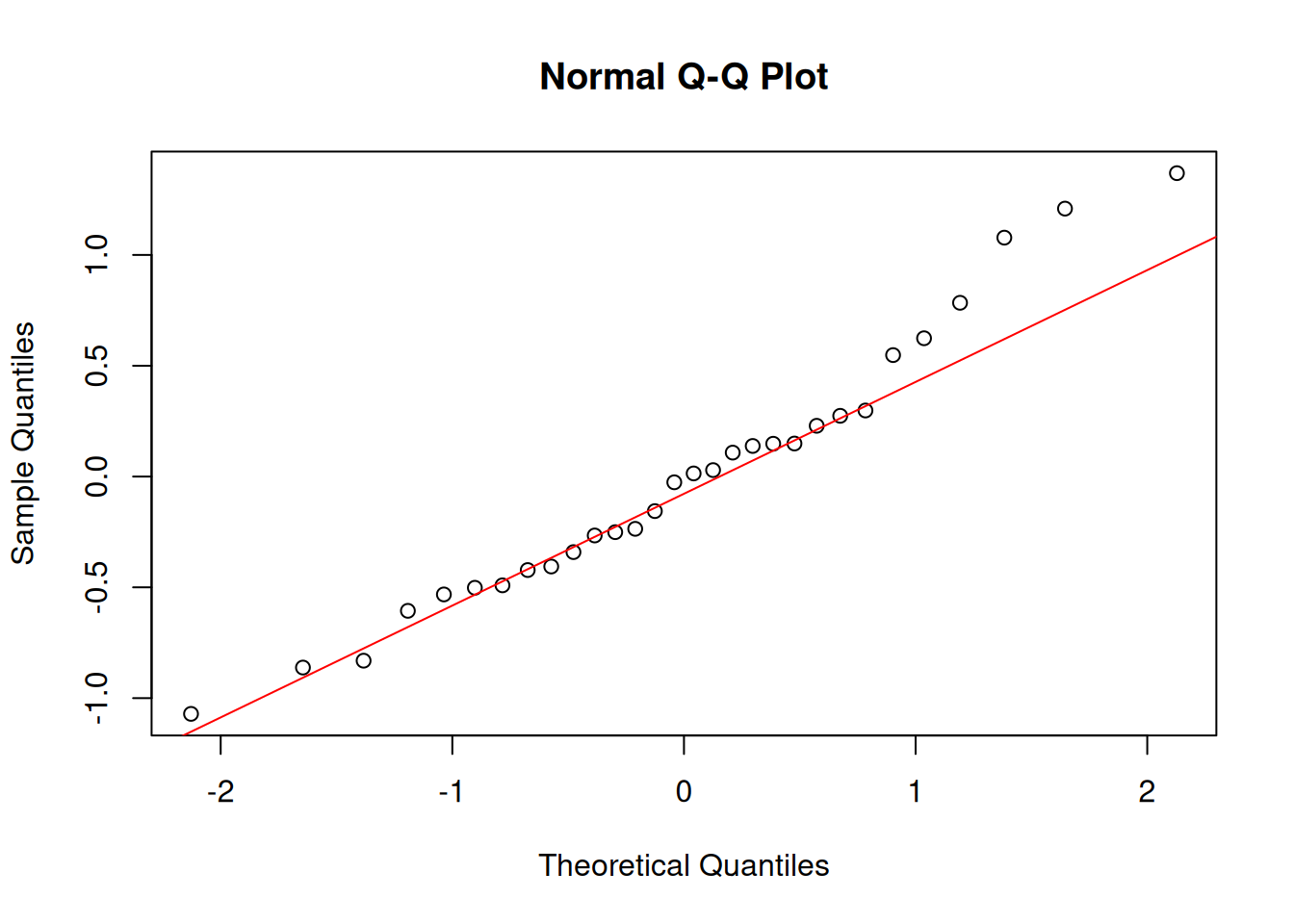
Always check residuals, not the raw data:
| Topic | Key functions |
|---|---|
| Objects & vectors | c(), class(), str(), summary(), head(), tail() |
| Type coercion | as.numeric(), as.character(), as.factor() |
| Data frames | data.frame(), $, [ , ], subset(), colSums(is.na()) |
| I/O | read.csv(), write.csv() |
| Iteration | apply(), sapply() |
| Pipe | \|> (base R ≥ 4.1) |
| ggplot2 | geom_point(), geom_histogram(), geom_density(), geom_smooth(), facet_wrap(), ggsave() |
| t-tests | t.test() (one-sample, two-sample, paired), wilcox.test() |
| Effect size | Cohen’s d (manual), eta_sq from aov() summary |
| ANOVA | aov(), TukeyHSD(), kruskal.test() |
| Multiple testing | p.adjust() (holm, fdr, bonferroni) |
| Correlation | cor.test() (pearson & spearman) |
| Categorical | fisher.test(), chisq.test() |
| Normality | shapiro.test(), qqnorm() |
Problem Set: R Stats Intro — due the Wednesday of Module 2. Submit on Canvas.
Explore concepts interactively (no R needed): Module 1 Interactive Explorers
---
title: "Module 1: R + Statistics Intro"
subtitle: "PLS 206 — Applied Multivariate Modeling"
author: "Grey Monroe"
date: "Fall 2026"
format:
html:
theme: flatly
toc: true
toc-depth: 3
toc-title: "Contents"
highlight-style: github
code-fold: false
fig-align: center
execute:
echo: true
warning: false
message: false
cache: false
---
# The Scientific Workflow
Everything in this course fits into one repeating cycle:
```
Collect data → Explore → Test / Model → Communicate
```
**This module** covers the first two steps — loading data and exploring it — plus the classical hypothesis tests you'll need when relationships are simple. **Modules 2–10** build the modeling toolkit for when they're not.
The single most important habit in data analysis: **look at your data before you model it.** Summary statistics can be identical across wildly different datasets (see Activity 3). Plot first, model second.
---
# Part 1: R Fundamentals
## Why R?
R is free, open-source, and used by ecologists, geneticists, agronomists, and data scientists worldwide. With 10,000+ packages on CRAN, it covers nearly every statistical method you'll need. More importantly, your entire analysis lives in a script — reproducible, shareable, and tweakable.
This quarter we'll use R for linear and generalized linear models, GAMs, regularization, PCA, clustering, random forests, neural nets, and SEM.
## Your First R Objects
```{r}
# Numeric
x <- 1
y <- 3.14
z <- 1e4 # scientific notation
# Math operations
x + y
(x + 1) * y # follows PEMDAS
sqrt(y)
log(y)
y^2
```
::: {.callout-note}
Use `<-` for assignment. Reserve `=` for function arguments only.
:::
## Data Types
```{r}
x <- 42 ; class(x) # numeric
w <- "hello" ; class(w) # character
flag <- TRUE ; class(flag) # logical
myvar <- 10
myvar == 10 # TRUE
myvar > 2 # TRUE
myvar < 2 # FALSE
```
### Type coercion
R will silently coerce types when you combine mismatched data — a common source of bugs when reading CSVs. Always check with `class()` or `str()` after import.
```{r}
# Convert between types explicitly
x_char <- "3.14"
class(x_char)
as.numeric(x_char) # "3.14" → 3.14
# The factor → numeric gotcha (one of R's most common traps)
f <- factor(c("10", "2", "30"))
as.numeric(f) # WRONG: returns factor level codes (1, 2, 3), not values!
as.numeric(as.character(f)) # CORRECT: go via character first
```
::: {.callout-warning}
**Never convert a factor directly to numeric.** Always go `as.numeric(as.character(x))`.
:::
## Vectors — the building block of R
Everything in R is a vector. A single number is a vector of length 1.
```{r}
numbers <- c(1, 2, 3, 4, 5, 10)
words <- c("apple", "banana", "cherry", "grape")
class(numbers)
length(numbers)
# Sequences
10:20
seq(from = 1, to = 100, by = 10)
```
## Vectors — operations & subsetting
```{r}
numbers * 2 # element-wise math
numbers[4:6] # index by position
words[2]
numbers[which(numbers > 3)] # index by condition
numbers[numbers > 3] # shorthand
words %in% c("apple", "grape") # membership test
words[words %in% c("apple", "grape")]
```
## Handling NAs and Factors
```{r}
# NA values propagate — use na.rm = TRUE
x <- c(1, 2, NA, 4)
is.na(x)
mean(x) # NA — because of the missing value
mean(x, na.rm = TRUE) # 2.333...
# Check for NAs across a data frame column-by-column
# colSums(is.na(df))
# Factors (categorical variables)
fruits <- factor(c("apple", "banana", "apple", "cherry"))
levels(fruits)
table(fruits)
```
## Lists & Data Frames
```{r}
# Lists hold mixed types
my_list <- list(number = 42, word = "hello", flag = TRUE)
my_list$number
my_list[["word"]]
# Data frames = the workhorse for your data
df <- data.frame(
rep = c(1, 2, 3, 4),
treat = c("A", "B", "A", "B"),
value = c(10.2, 8.5, 11.1, 9.3)
)
str(df)
```
## Working with Data Frames
```{r}
df$value # access a column
df[1, ] # first row
df[df$treat == "A", ] # filter rows
# Quick looks at your data
head(df) # first 6 rows (default)
tail(df) # last 6 rows — good for checking import completeness
head(df, 10) # first 10 rows
# Add a column
df$log_value <- log(df$value)
summary(df)
```
## Reading and Writing Data
```{r}
wine <- read.csv("winedata.csv")
str(wine)
head(wine)
```
::: {.callout-tip}
Always run `str()` after reading data — verify that column types match what you expect (e.g., numeric not character).
:::
```{r}
#| eval: false
# Save a modified data frame
write.csv(wine, "winedata_clean.csv", row.names = FALSE)
```
## The Pipe Operator
You will see `|>` (base R pipe, R ≥ 4.1) and `%>%` (magrittr, from the tidyverse) constantly in R code online. They both mean the same thing: **pass the left-hand side as the first argument to the right-hand side.**
```{r}
# These three are identical:
mean(c(1, 2, 3, NA), na.rm = TRUE)
c(1, 2, 3, NA) |> mean(na.rm = TRUE)
# Reading it aloud: "take this vector, THEN compute the mean"
```
Pipes shine when you chain multiple operations:
```{r}
# Without pipe — reads inside-out
round(sqrt(abs(-16)), 2)
# With pipe — reads left to right
-16 |> abs() |> sqrt() |> round(2)
```
::: {.callout-note}
`|>` is built into R ≥ 4.1 — no packages needed. `%>%` requires `library(magrittr)` or `library(dplyr)`. They behave identically in most cases. Prefer `|>` in new code.
:::
## Iteration: apply and loops
When you need to do the same operation across many columns or groups, reach for `apply()` or `sapply()` instead of copy-pasting code. This will come up immediately in the problem set.
```{r}
data(iris)
# apply() — loops over rows (MARGIN=1) or columns (MARGIN=2) of a matrix/data frame
apply(iris[, 1:4], MARGIN = 2, FUN = mean) # mean of each numeric column
# sapply() — loops over a vector or list, returns a simplified result
sapply(iris[, 1:4], sd) # SD of each numeric column
# Equivalent for loop (more verbose, same result)
for (col in names(iris)[1:4]) {
cat(col, "mean =", mean(iris[[col]]), "\n")
}
```
::: {.callout-note}
More on `apply()`, `lapply()`, and writing your own functions comes in Module 2. For now, the pattern is: `apply(data_frame, 2, some_function)`.
:::
---
# Part 1b: ggplot2
## The Grammar of Graphics
`ggplot2` builds plots layer by layer:
```
ggplot(data, aes(x = ..., y = ...)) +
geom_*() + # geometry: points, lines, bars, histograms ...
labs() + # labels
theme_*() # styling
```
Every plot starts with `ggplot()`. Add layers with `+`.
## Distributions: Histogram and Density
The first thing to do with any new variable — look at its distribution.
```{r}
library(ggplot2)
# Histogram
ggplot(iris, aes(x = Sepal.Length)) +
geom_histogram(bins = 20, fill = "steelblue", color = "white") +
labs(x = "Sepal Length (mm)", y = "Count") +
theme_classic()
```
```{r}
# Density curve — smooth estimate of the distribution
ggplot(iris, aes(x = Sepal.Length, fill = Species)) +
geom_density(alpha = 0.4) +
labs(x = "Sepal Length (mm)", y = "Density") +
theme_classic()
```
## Scatter Plot
```{r}
ggplot(iris, aes(x = Sepal.Length, y = Sepal.Width)) +
geom_point()
```
## Add a Fit Line
```{r}
ggplot(iris, aes(x = Sepal.Length, y = Sepal.Width)) +
geom_point() +
geom_smooth(method = "lm")
```
## Color by Group
```{r}
ggplot(iris, aes(x = Sepal.Length, y = Sepal.Width, col = Species)) +
geom_point() +
geom_smooth(method = "lm")
```
## Faceting
`facet_wrap()` splits one plot into panels by a grouping variable — one of ggplot's most powerful tools for exploratory analysis.
```{r}
ggplot(iris, aes(x = Sepal.Length, y = Petal.Length)) +
geom_point(alpha = 0.6) +
geom_smooth(method = "lm", se = FALSE) +
facet_wrap(~ Species) +
theme_classic()
```
## Publication-Ready Plot
```{r}
ggplot(iris,
aes(x = Sepal.Length, y = Sepal.Width,
col = Species, fill = Species)) +
geom_point(alpha = 0.7) +
geom_smooth(method = "lm", alpha = 0.1) +
labs(title = "Iris Dataset",
x = "Sepal Length (mm)",
y = "Sepal Width (mm)") +
scale_color_manual(values = c("gray30", "dodgerblue", "orange")) +
scale_fill_manual(values = c("gray30", "dodgerblue", "orange")) +
theme_classic(base_size = 14) +
theme(plot.title = element_text(face = "bold"),
legend.position = "top")
```
## Saving Plots
```{r}
#| eval: false
# Save the last plot as a PDF (preferred for publication)
ggsave("sepal_scatter.pdf", width = 5, height = 4)
# Or save a named plot object
p <- ggplot(iris, aes(Sepal.Length, Sepal.Width)) + geom_point()
ggsave("sepal.png", plot = p, width = 5, height = 4, dpi = 300)
```
::: {.callout-tip}
Use `ggsave()` rather than screenshot. It gives you a reproducible, high-resolution file at any dimension you need.
:::
::: {.callout-note}
## Exploring many variables at once: GGally
The `GGally` package extends ggplot2 with `ggpairs()` — a scatter plot matrix showing pairwise relationships, distributions, and correlations in one figure. You'll use it in the problem set:
```r
library(GGally)
ggpairs(wine_reduce, columns = 2:7) # all pairs
ggpairs(wine_reduce, aes(col = Cultivar), columns = 2:7) # colored by group
```
:::
---
# Part 2: Statistics Fundamentals
## What is a p-value? (and what isn't it)
Before running any tests, anchor the key concepts.
A **p-value** is the probability of observing a result at least this extreme *if the null hypothesis were true*. It is **not**:
- The probability that the null hypothesis is true
- The probability that your result is a false positive
- A measure of effect size or practical importance
- Proof of anything
::: {.callout-warning}
**p < 0.05 does not mean the effect is real or important.** With n = 10,000, a trivially small effect will be significant. Always report *both* a p-value and an effect size.
:::
### Type I and Type II errors
| | H₀ is actually true | H₀ is actually false |
|---|---|---|
| **Reject H₀** (p < α) | Type I error (false positive) — rate = α | Correct (true positive) — power = 1 − β |
| **Fail to reject H₀** | Correct (true negative) | Type II error (false negative) — rate = β |
Setting α = 0.05 means you accept a 5% chance of a false positive **per test** — which becomes a serious problem when running many tests (see [Multiple Comparisons](#multiple-comparisons) below).
**Statistical power** (1 − β) is the probability of detecting a real effect. Power increases with larger sample size, larger effect size, and lower measurement noise. See the [t-test Explorer](Mod1_interactive.html) to watch power change in real time.
## Choosing the Right Test
| Outcome | Groups / structure | Normally distributed? | Test |
|---|---|---|---|
| Continuous | 1 group vs. fixed value | Yes | One-sample t-test |
| Continuous | 1 group vs. fixed value | No | Wilcoxon signed-rank |
| Continuous | 2 independent groups | Yes | Welch's two-sample t-test |
| Continuous | 2 independent groups | No | Mann-Whitney U |
| Continuous | 2 paired / matched groups | Yes | Paired t-test |
| Continuous | 2 paired / matched groups | No | Wilcoxon signed-rank (paired) |
| Continuous | ≥ 3 independent groups | Yes | One-way ANOVA + TukeyHSD |
| Continuous | ≥ 3 independent groups | No | Kruskal-Wallis + pairwise Wilcoxon |
| Continuous | 2 continuous variables | Linear | Pearson correlation |
| Continuous | 2 continuous variables | Monotone / outliers | Spearman correlation |
| Categorical | 2 × 2 table, small n | — | Fisher's exact |
| Categorical | 2 × 2 or larger, large n | — | Chi-squared |
::: {.callout-tip}
**Normality** refers to your *residuals*, not your raw data. For small samples (n < 30), check with `shapiro.test()` and a QQ plot. For large samples, the central limit theorem means most tests are robust even with non-normal data.
:::
## Descriptive Statistics
```{r}
set.seed(42)
data("PlantGrowth")
tapply(PlantGrowth$weight, PlantGrowth$group, summary)
```
```{r}
ggplot(PlantGrowth, aes(group, weight, fill = group)) +
geom_boxplot(show.legend = FALSE, alpha = 0.7) +
geom_jitter(width = 0.08, alpha = 0.5) +
labs(x = "Treatment", y = "Weight (g)") +
theme_classic()
```
## One-Sample t-test
Does the control group mean differ from 5.0 g?
```{r}
ctrl <- subset(PlantGrowth, group == "ctrl")$weight
t.test(ctrl, mu = 5)
```
Non-parametric alternative:
```{r}
wilcox.test(ctrl, mu = 5, conf.int = TRUE)
```
## Two-Sample t-test
```{r}
x1 <- subset(PlantGrowth, group == "ctrl")$weight
x2 <- subset(PlantGrowth, group == "trt1")$weight
# Welch's t-test (default — allows unequal variances)
t.test(x1, x2)
```
```{r}
# Mann-Whitney U (non-parametric)
wilcox.test(x1, x2, conf.int = TRUE)
```
::: {.callout-tip}
**Welch's t-test** is the safe default — it doesn't assume equal variances between groups.
:::
## Paired t-test
Use a paired t-test when each observation in group A has a natural match in group B — before/after measurements on the same subject, plots receiving both treatments, twin studies. Pairing removes between-subject variation and increases power.
```{r}
# Simulated example: soil nitrate (mg/kg) at 12 plots before and after treatment
set.seed(42)
pre <- rnorm(12, mean = 10, sd = 2)
post <- pre + rnorm(12, mean = 2.5, sd = 1.5) # treatment adds ~2.5 mg/kg
# Paired t-test
t.test(post, pre, paired = TRUE)
# Equivalent: one-sample t-test on the differences
t.test(post - pre, mu = 0)
```
```{r}
# Visualize paired structure
plot_df <- data.frame(
plot = rep(1:12, 2),
time = rep(c("Pre", "Post"), each = 12),
nitrate = c(pre, post)
)
plot_df$time <- factor(plot_df$time, levels = c("Pre", "Post"))
ggplot(plot_df, aes(x = time, y = nitrate, group = plot)) +
geom_line(alpha = 0.5) +
geom_point(size = 2) +
labs(x = NULL, y = "Nitrate (mg/kg)",
title = "Paired before/after measurements") +
theme_classic()
```
Non-parametric alternative:
```{r}
wilcox.test(post, pre, paired = TRUE, conf.int = TRUE, exact = FALSE)
```
## Effect Size
A significant p-value tells you the effect probably isn't zero. **Effect size** tells you how big it is — which is what actually matters for biology.
### Cohen's d (for t-tests)
$$d = \frac{|\bar{x}_1 - \bar{x}_2|}{s_\text{pooled}}$$
```{r}
# Cohen's d for the two-sample comparison above
pooled_sd <- sqrt((var(x1) + var(x2)) / 2)
cohens_d <- abs(mean(x1) - mean(x2)) / pooled_sd
cohens_d
```
| d | Interpretation |
|---|---|
| < 0.2 | Negligible |
| 0.2 – 0.5 | Small |
| 0.5 – 0.8 | Medium |
| > 0.8 | Large |
### η² (eta-squared) for ANOVA
η² is the proportion of total variance explained by the group factor — analogous to R² in regression.
```{r}
model <- aov(weight ~ group, data = PlantGrowth)
ss <- summary(model)[[1]][["Sum Sq"]]
eta_sq <- ss[1] / sum(ss) # SS_group / SS_total
eta_sq
```
::: {.callout-tip}
Report effect sizes in your results. A biologically trivial effect can be highly significant with a large enough sample. A large effect can be non-significant with a small sample. Neither alone is the whole story.
:::
## One-Way ANOVA
Do all three treatment groups differ?
```{r}
model <- aov(weight ~ group, data = PlantGrowth)
summary(model)
```
Post-hoc pairwise comparisons:
```{r}
TukeyHSD(model)
```
## ANOVA vs Kruskal-Wallis
```{r}
#| layout-ncol: 2
ggplot(PlantGrowth, aes(group, weight, fill = group)) +
geom_boxplot(alpha = 0.75, show.legend = FALSE) +
geom_jitter(width = 0.08, alpha = 0.6) +
labs(title = "Raw values (ANOVA)") +
theme_classic()
ggplot(PlantGrowth, aes(group, rank(weight), fill = group)) +
geom_boxplot(alpha = 0.75, show.legend = FALSE) +
geom_jitter(width = 0.08, alpha = 0.6) +
labs(title = "Ranks (Kruskal-Wallis)") +
theme_classic()
```
```{r}
kruskal.test(weight ~ group, data = PlantGrowth)
```
## Multiple Comparisons {#multiple-comparisons}
Every statistical test you run has a α = 0.05 chance of a false positive. If you run 20 independent tests, you expect **one false positive on average** — not because the biology changed, but because of random chance.
::: {.callout-warning}
**The problem:** computing 10 pairwise correlations between wine variables gives you 10 chances for a spurious significant result. Running ANOVA post-hoc tests compounds the error further.
**The solution:** adjust p-values upward to account for the number of tests.
:::
Common methods, roughly in order of conservatism:
- **Holm** (default in `pairwise.wilcox.test`, `p.adjust`) — good general choice, less conservative than Bonferroni
- **Bonferroni** — multiply each p by the number of tests; very conservative
- **FDR / Benjamini-Hochberg** — controls the *false discovery rate*; preferred when running hundreds of tests (e.g., genomics)
```{r}
# Pairwise Wilcoxon with Holm correction
pairwise.wilcox.test(PlantGrowth$weight,
PlantGrowth$group,
p.adjust.method = "holm")
# Manually adjust a vector of p-values
p_raw <- c(0.01, 0.04, 0.03, 0.20, 0.005)
p.adjust(p_raw, method = "holm")
p.adjust(p_raw, method = "fdr")
```
## Pearson vs Spearman Correlation
```{r}
data("CO2")
sub <- subset(CO2, Type == "Quebec" & Treatment == "nonchilled")
cor.test(sub$conc, sub$uptake, method = "pearson")
cor.test(sub$conc, sub$uptake, method = "spearman", exact = FALSE)
```
```{r}
#| layout-ncol: 2
ggplot(sub, aes(conc, uptake)) +
geom_point() +
geom_smooth(method = "lm", linewidth = 0.8) +
geom_smooth(method = "loess", linetype = 2, linewidth = 0.8) +
labs(title = "Raw", x = "CO2 concentration", y = "Uptake") +
theme_classic()
ggplot(sub, aes(rank(conc), rank(uptake))) +
geom_point() +
geom_smooth(method = "lm", linewidth = 0.8) +
labs(title = "Ranked (Spearman)", x = "Rank", y = "Rank") +
theme_classic()
```
## Categorical Data — Fisher's Exact & Chi-Squared
Is pest presence associated with treatment?
```{r}
tab <- matrix(c(3, 17, 10, 10), nrow = 2, byrow = TRUE,
dimnames = list(Pest = c("Present","Absent"),
Trt = c("A","B")))
tab
fisher.test(tab)
chisq.test(tab, correct = FALSE)
```
::: {.callout-tip}
Use **Fisher's exact** when any cell count is < 5. Use **chi-squared** for larger samples.
:::
## Checking Normality
Always check residuals, not the raw data:
```{r}
#| layout-ncol: 2
model <- aov(weight ~ group, data = PlantGrowth)
resids <- residuals(model)
hist(resids, main = "ANOVA residuals", col = "steelblue")
qqnorm(resids); qqline(resids, col = "red")
```
```{r}
shapiro.test(resids)
```
---
# Module 1 Summary
| Topic | Key functions |
|---|---|
| Objects & vectors | `c()`, `class()`, `str()`, `summary()`, `head()`, `tail()` |
| Type coercion | `as.numeric()`, `as.character()`, `as.factor()` |
| Data frames | `data.frame()`, `$`, `[ , ]`, `subset()`, `colSums(is.na())` |
| I/O | `read.csv()`, `write.csv()` |
| Iteration | `apply()`, `sapply()` |
| Pipe | `\|>` (base R ≥ 4.1) |
| ggplot2 | `geom_point()`, `geom_histogram()`, `geom_density()`, `geom_smooth()`, `facet_wrap()`, `ggsave()` |
| t-tests | `t.test()` (one-sample, two-sample, paired), `wilcox.test()` |
| Effect size | Cohen's d (manual), `eta_sq` from `aov()` summary |
| ANOVA | `aov()`, `TukeyHSD()`, `kruskal.test()` |
| Multiple testing | `p.adjust()` (holm, fdr, bonferroni) |
| Correlation | `cor.test()` (pearson & spearman) |
| Categorical | `fisher.test()`, `chisq.test()` |
| Normality | `shapiro.test()`, `qqnorm()` |
**Problem Set: R Stats Intro** — due the Wednesday of Module 2. Submit on Canvas.
---
Explore concepts interactively (no R needed): **[Module 1 Interactive Explorers](Mod1_interactive.html)**